TransCon PTH
Designed to replace parathyroid hormone to physiologic levels, 24 hours a day, every day, for patients with hypoparathyroidism
Designed to replace parathyroid hormone to physiologic levels, 24 hours a day, every day for patients with hypoparathyroidism
Parathyroid hormone (PTH) plays a critical role in controlling calcium, phosphate and calcitriol (active vitamin D) levels in the blood and bone. Through its primary actions, PTH directly involves the kidneys, bone and other organs in maintaining key biological functions.
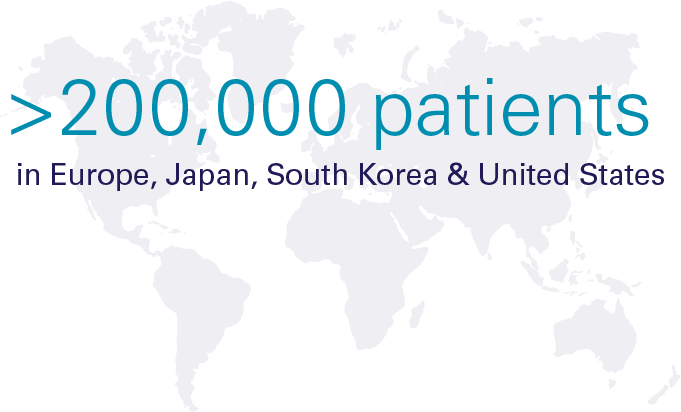
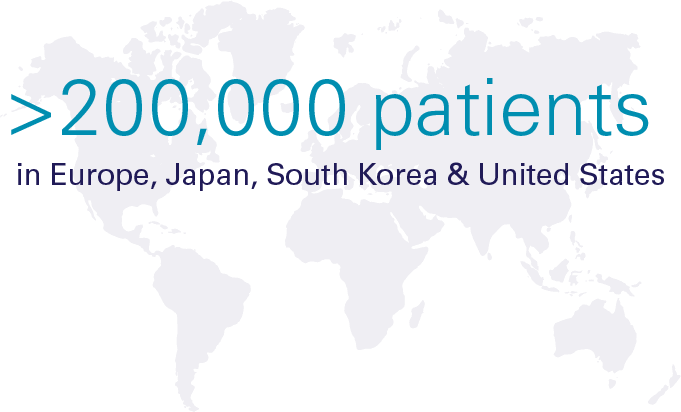
Hypoparathyroidism (hypopara or HP) is a rare endocrine disorder characterized by deficient or absent parathyroid hormone. It affects over 200,000 people worldwide, most of whom develop the condition following damage to or accidental removal of the parathyroid glands during thyroid surgery. Current standard of care treatment with calcium and calicitriol (or the analogue alfacalcidol/alphacalcidol) does not effectively address both the short-term symptoms and long-term complications, or the quality-of-life impacts of hypoparathyroidism.
Significant Global Prevalence
Causes of Hypopara
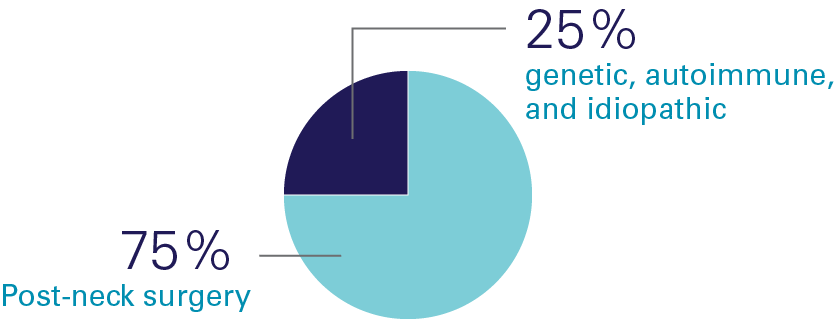
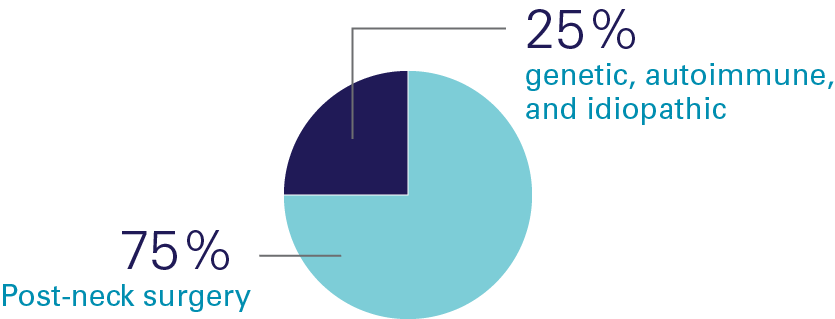
Burden of disease
People living with hypoparathyroidism experience a diverse range of physical, mental, cognitive and emotional symptoms. They often suffer debilitating symptoms and the condition can severely impact quality of life and overall productivity, including ability to work and perform household activities.
Even worse, the current standard of care with supplementation of calcium and calicitriol or the analogue alfacalcidol/alphacalcidol does not fully control the condition, poses a significant daily pill burden, and contributes to risk of long-term complications, including renal disease. People with hypoparathyroidism have at least a four-fold greater risk of kidney failure than the general population, in part due to excessive urinary calcium excretion and long-term complications caused by standard treatment.
Hypoparathyroidism is also linked to increased hospitalizations and emergency room and urgent care visits. In some cases, calcium levels fall so low that patients experience a ‘calcium crash’ and urgently require calcium delivered intravenously – a situation often dreaded by patients, their friends and family.
A survey of people living with hypoparathyroidism (N=146) showed patients required multiple physician visits and up to one decade to diagnose their condition.
A complex patient experience
![]()
Hypocalcemia (low calcium in the blood)
- Abnormal tingling, burning, numbness (paresthesia)
- Brain fog (impaired judgement, memory loss, headache)
- Muscle cramps (tetany)
- Seizures
- Laryngospasm (spasm of the voice box)
Hypercalcemia (too much calcium in the blood)
- Muscle weakness, coma, constipation, urination difficulties
Anxiety/fear
- May relate to potential calcium crash
![]()
Calcification
- Calcium deposits in the kidney, brain, blood vessels, eye and other soft tissues
Kidney disease
- 4- to 8-fold increased risk of kidney disease compared to healthy populations
Depression or bipolar disorder
- 2-fold increased risk of depression or bipolar disorder compared to healthy populations
![]()
Negative psychological impacts
- 100% of patients said their condition interferes with their daily lives
Impacts on physical functioning
- 85% reported an inability to perform household activities
Negative impacts on the ability to work or work productivity
- 76% reported their condition interfered with work productivity
- 30% were no longer able to work
TransCon PTH
TransCon® is an investigational long-acting prodrug of parathyroid hormone (PTH) in development as a once-daily hormone replacement therapy. It is designed to restore physiologic levels of PTH 24 hours a day. By doing so, we believe TransCon PTH can address both the short-term symptoms and long-term complications of hypoparathyroidism. Our goal is to provide a PTH replacement therapy that normalizes serum and urinary calcium, as well as serum phosphate levels, and improves quality of life. TransCon PTH has orphan designation for the treatment of hypoparathyroidism in both the U.S. and Europe.
Program Status
The PaTHway Trial is a double-blind, placebo-controlled trial evaluating TransCon PTH as a treatment in adults with hypoparathyroidism. The global trial is expected to enroll approximately 76 participants at sites in North America and Europe. TransCon PTH is stable at room temperature and administered in a ready-to-use liquid formulation with a pen-injector.
In 2020, we reported results of PaTH Forward, a phase 2, randomized, double-blind, placebo-controlled, parallel group trial evaluating the safety and efficacy of TransCon PTH in adults with hypoparathyroidism. Results from the trial demonstrated that TranCon PTH has the potential to eliminate standard treatment with oral calcium, and oral calcitriol or its analogues, while normalizing serum and urinary calcium levels, as well as serum phosphate levels, and may improve quality of life. Participants continue to be evaluated in the long-term, open-label portion of the trial.