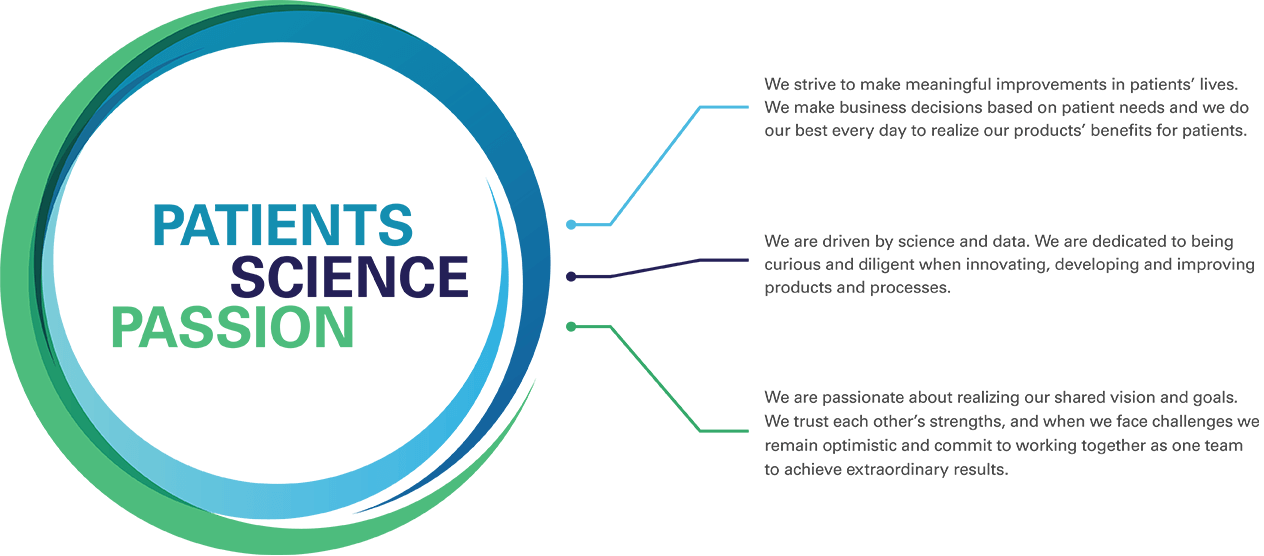
We are applying our innovative technology platform to build a leading, fully integrated global biopharmaceutical company
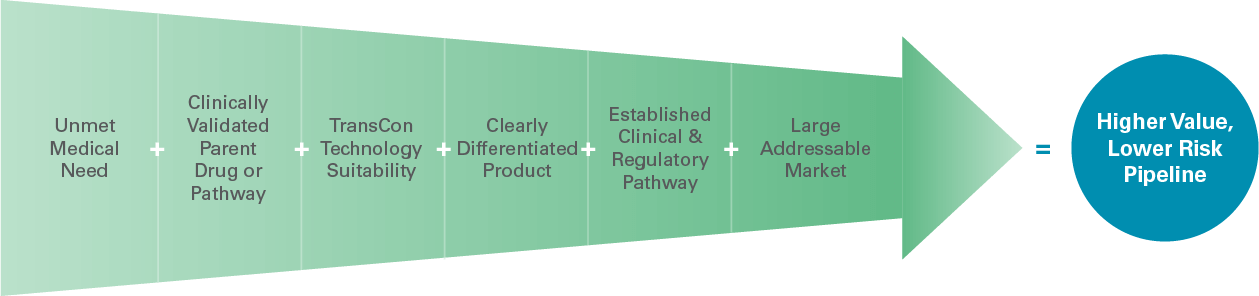
Algorithm for product innovation
Our unique approach to product innovation focuses on identifying unmet medical needs where a clinically validated parent drug or pathway is suitable to our TransCon technologies. We call this our “algorithm.” It helps us select and advance product candidates for our ever-growing pipeline.
Leveraging this strategic approach, we currently have a pipeline of three independent endocrinology rare disease product candidates in clinical development and we are advancing oncology as a second therapeutic area of focus. We continue to expand into new therapeutic areas to address unmet patient needs.
Our vision is to create new therapies focused on making a meaningful difference in patients’ lives.
VISEN Pharmaceuticals
In 2018, VISEN Pharmaceuticals was formed by Ascendis Pharma with an investor syndicate led by Vivo Capital. Focusing on endocrinology, VISEN has received exclusive rights to develop and commercialize Ascendis Pharma's endocrinology rare disease therapies, including TransCon hGH, TransCon PTH and TransCon CNP in Greater China.